Otolaryngologist, head and neck surgeon Francis T. Hall discusses the evaluation of thyroid nodules, which primarily aims to determine the likelihood of malignancy. He then reviews the treatment of thyroid nodules and thyroid cancer, including recent advances in management
Radiology plays many roles in prostate cancer, and novel therapies are emerging
Radiology plays many roles in prostate cancer, and novel therapies are emerging
We are on our summer break and the editorial office is closed until 17 January. In the meantime, please enjoy our Summer Hiatus series, an eclectic mix from our news and clinical archives and articles from The Conversation throughout the year. This article was first published in the 15 September edition
To coincide with Prostate Cancer Foundation NZ’s Blue September, here is a case study that illustrates the use of radiology and radioligand therapy across a prostate cancer journey
Remy Lim
- Multiparametric prostate MRI shows exquisite details of the prostate gland and is an essential first step in diagnosing prostate cancer.
- Prostate MRI enables superior detection of clinically significant cancers compared with biopsy of the prostate gland; it can also be used to target high-risk abnormalities for biopsy or to look for local recurrence after surgery.
- A prostate-specific membrane antigen PET/CT scan is useful for staging and restaging of prostate cancer as it is excellent in diagnosing early metastasis in lymph nodes and bone.
- Lutetium-177 PSMA radioligand therapy is an emerging and promising therapy to palliate men with advanced metastatic prostate cancer.
This article has been endorsed by the RNZCGP and has been approved for up to 0.25 CME credits for the General Practice Educational Programme and continuing professional development purposes (1 credit per learning hour). To claim your credits, log in to your RNZCGP dashboard to record this activity in the CME component of your CPD programme.
Nurses may also find that reading this article and reflecting on their learning can count as a professional development activity with the Nursing Council of New Zealand (up to 0.25 PD hours).
Metastatic disease outside the prostate bed…may be targeted for localised radiation if isolated
Andy is a 52-year-old man who comes in requesting a “prostate check” due to a strong family history of prostate cancer – an older brother was diagnosed at age 56, and their father was diagnosed with prostate cancer at age 74. He has no lower urinary tract symptoms and declines a digital rectal examination.
You have a robust discussion about the pros and cons of checking his prostate-specific antigen (PSA) and oblige by ticking PSA on the blood form. His PSA level comes back at 4.8ng/ml, and a repeat PSA test also comes back elevated at 5.0ng/ml. You decide to refer Andy to a urologist for review and further management.
Prostate MRI
Elevated PSA is the most common clinical indication for men undergoing a multiparametric MRI (mpMRI) of the prostate gland, which is an essential first step in diagnosing prostate cancer.
Advances in MRI technology over the last 10 years and widespread availability of 3 Tesla scanners allow us to see exquisite details of the prostate gland. Due to the superior soft tissue contrast resolution with MRI, the different zones of the prostate gland can usually be clearly delineated, making it a valuable imaging tool compared with other modalities such as ultrasound or CT scan.
Anatomically, the prostate gland consists of a peripheral zone, transition zone, central zone and the anterior fibromuscular stroma. Approximately 70 per cent of prostate cancers arise in the peripheral zone, which is partially accessible with digital rectal examination. The transition zone is commonly the site of benign prostatic hyperplasia, but the transition zone may also harbour malignancy – up to 15 per cent of prostate cancers. It is relatively uncommon to diagnose a central zone tumour.
Better understanding of the biology of prostate cancer in recent years has shown that we can safely monitor some low-grade prostate cancers. Active treatment is only indicated for cancers that are “clinically significant” – with a Gleason score of 7, or International Society of Urological Pathology (ISUP) grade 2, or above.
What data do we have to support the use of MRI to evaluate clinically significant prostate cancers? There have been two landmark studies in the last four years that have put MRI at the forefront of prostate cancer diagnosis.
The PROMIS study compared detection accuracy of mpMRI and transrectal ultrasound-guided (TRUS) biopsy of the prostate gland against the gold standard reference of transperineal template-guided mapping (TPM) biopsy, which involves extensive biopsy of the entire prostate gland.1
Of the 576 patients enrolled in this study, 40 per cent had clinically significant cancer on TPM biopsy. MRI significantly outperformed TRUS biopsy in diagnosing clinically significant cancer with a sensitivity of 93 per cent versus 48 per cent. Further, by performing mpMRI in those with elevated PSA, the study shows one-quarter of men can avoid the need for a prostate biopsy altogether.1
Hot on the heels of PROMIS was the PRECISION study, which showed clinically significant cancers are more likely to be detected with mpMRI-targeted biopsy than with TRUS biopsy. Additionally, by performing mpMRI risk assessment beforehand, fewer low-risk cancers (Gleason 6 or ISUP 1) are diagnosed at biopsy, which is also a good thing for patients.2
So, what does mpMRI of the prostate gland consist of? The word multiparametric simply refers to employing multiple different “acquisitions” or “sequences” to increase the conspicuity of a potentially cancerous lesion in the prostate gland. There may be up to eight different sequences in a prostate MRI examination, mostly in the axial plane, resulting in an examination time of up to 30–40 minutes.
The different sequences are often referred to as T1 or T2 weighted, but the most crucial sequence to get right in prostate MRI is the diffusion-weighted imaging sequence. This detects “restriction” in the random Brownian motion of water molecules, which tends to occur when cancerous cells are tightly packed together.
Integration of these various sequences, solid knowledge of cancer characteristics and experience in reporting prostate MRI are all essential in ensuring clinically significant cancers are diagnosed and treated in a timely fashion.
On a prostate MRI report, you will also most likely see a prostate imaging reporting and data system (PI-RADS) score, which ranges from 1 to 5. PI-RADS 1 is essentially a normal examination, but a PI-RADS 4 or 5 denotes a lesion highly suspicious for a cancer based on the signal characteristics of the lesion, size and/or presence of extracapsular extension.
Andy is found to have a PI-RADS 4 lesion in the left lateral peripheral zone, meaning there is high likelihood of a clinically significant cancer. He has a prostate volume of 32cc, so his PSA density (PSA/prostate volume) of 0.16ng/ml/cc is also borderline elevated. His urologist recommends a transperineal biopsy to target the lesion of concern.
PSMA PET/CT imaging
Unfortunately, Andy’s biopsy returns a positive diagnosis of Gleason 8 or ISUP 4 disease. He consults his urologist on the possibility of radical prostatectomy and is also referred to a radiation oncologist for consideration of radiation therapy. He chooses to undergo radical prostatectomy.
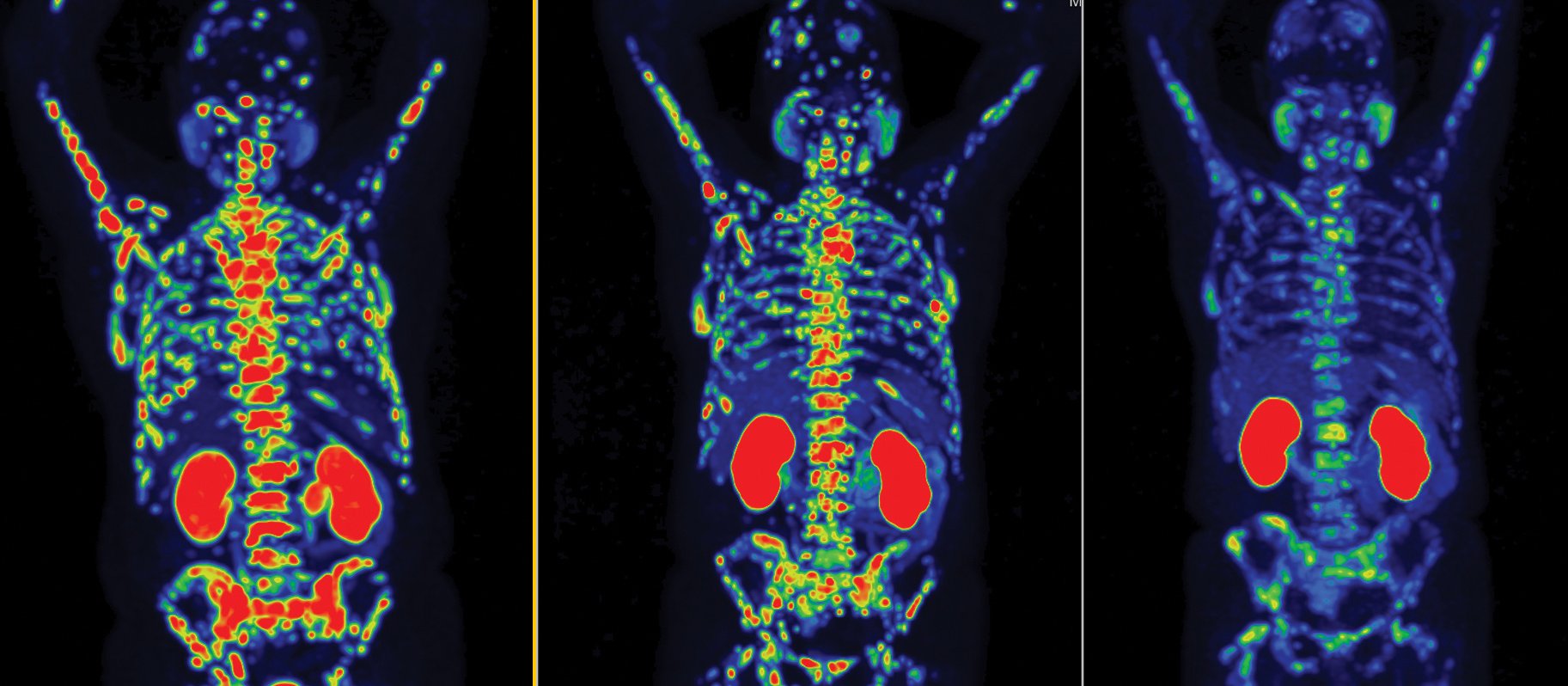
Due to his high-risk disease, the urologist feels Andy would be best served with a prostate-specific membrane antigen (PSMA) positron emission tomography (PET)/CT staging scan before his surgery. This demonstrates disease within the prostate gland concordant with the MRI findings. Fortunately, the cancer remains organ-confined without regional nodal or distant metastatic disease.
PSMA is a glycopeptide found on 90 per cent of prostate cancer cells. Although the name implies it is specific to prostate cancer, physiological or non-malignant PSMA expression is also found in salivary glands, the proximal small bowel, spleen, and the neovasculature of a host of non-prostatic cancers. Nevertheless, a PSMA PET/CT scan is excellent in diagnosing early metastasis in normal (morphologically and size wise) lymph nodes. It is also highly sensitive in detecting bony metastasis.
The proPSMA randomised controlled study from Australia showed PSMA PET/CT had superior sensitivity and specificity when compared with conventional imaging using CT and bone scan in staging of patients with prostate cancer.3
Further, an Auckland study showed that performing the scan before surgery has the potential to change management in up to 25 per cent of patients, avoiding futile surgery in cases where metastatic disease may otherwise be occult on conventional imaging.4
Lutetium-177 PSMA radioligand therapy
Andy undergoes a successful radical prostatectomy, and PSA remains undetectable for the first two years. However, a routine evaluation two and a half years post-operatively detects a rising PSA level.
In this scenario, a repeat MRI of the prostatectomy fossa to look for local recurrence is often quite useful if salvage radiation to the prostate bed is an option.
PSMA PET/CT is also useful to evaluate for metastatic disease outside the prostate bed, such as in pelvic lymph nodes or skeleton, as these may be targeted for localised radiation if isolated, potentially sparing the patient from needing to start hormone therapy straight away.
Andy’s PSMA PET/CT and pelvic MRI demonstrate metastatic disease in a couple of pelvic lymph nodes as well as local disease recurrence. He is commenced on androgen deprivation therapy and undergoes salvage radiotherapy to the prostate bed and lymph nodes. The reduction in PSA is short-lived, and after several lines of treatment over 18 months, he becomes castrate resistant, with rising PSA despite maximal androgen blockade.
His disease is controlled for a short period with docetaxel chemotherapy but then progresses rapidly after six cycles of chemotherapy and develops into progressive metastatic bone disease.
Andy is recommended to consider an emerging and promising palliative treatment called lutetium-177 PSMA radioligand therapy, one of the last few options available for palliation, with a response rate of 50 per cent in men who have been heavily pretreated with other therapy.
Lu-177 is an emitter of beta particles and can be coupled to a ligand that seeks out the same PSMA peptides detected on PSMA PET/CT scans. Once the ligand attaches to the PSMA peptides, the emission of beta particles causes DNA damage to the cancer cells and, ultimately, cell death.
The recent phase III VISION trial demonstrated superiority of Lu-PSMA radioligand therapy over standard of care in terms of survival benefit.5 Further, a recent phase II trial has shown the treatment to be “gentler” compared with second-line chemotherapy cabizataxel, with higher PSA response and fewer grade 3 or 4 adverse events.6
The Lu-PSMA radioligand therapy is successful in controlling Andy’s disease for a further 12 months, reducing his PSA and alleviating some of his bone pain. Two restaging PSMA PET/CT scans show radiographic response. However, as is the nature of the disease, Andy lives for another eight months with best supportive care and eventually succumbs five years after diagnosis.
Andy’s journey is an illustration of the roles radiology plays in the diagnosis, staging and restaging of prostate cancer. Lu-PSMA radioligand therapy brings a new paradigm to advanced metastatic prostate cancer, combining the “looking” with “treating” in a palliative setting.
Remy Lim is the medical director for Mercy Radiology and a radiologist at Auckland City Hospital
Details have been changed to protect patient confidentiality
You can use the Capture button below to record your time spent reading and your answers to the following learning reflection questions:
- Why did you choose this activity (how does it relate to your PDP learning goals)?
- What did you learn?
- How will you implement the new learning into your daily practice?
- Does this learning lead to any further activities that you could undertake (audit activities, peer discussions, etc)?
We're publishing this article as a FREE READ so it is FREE to read and EASY to share more widely. Please support us and our journalism – subscribe here
1. Ahmed HU, El-Shater Bosaily A, Brown LC, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 2017;389(10071):815–22.
2. Kasivisvanathan V, Rannikko AS, Borghi M, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 2018;378(19):1767–77.
3. Hofman MS, Lawrentschuk N, Francis RJ, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet 2020;395(10231):1208–16.
4. Tarr GP, Kashyap P, Dixit DD, et al. Utility of Ga68 prostate-specific membrane antigen positron-emission tomography for pre-operative staging of high-risk prostate cancer in a prospective cohort. J Med Imaging Radiat Oncol 2020;64(1):78–86.
5. Sartor O, de Bono J, Chi KN, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med 2021;10.1056/NEJMoa2107322.
6. Hofman MS, Emmett L, Sandhu S, et al. [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet 2021;397(10276):797–804.





![New Zealand Doctor Rata Aotearoa editor Barbara Fountain, RNZCGP president elect and Tauranga-based specialist GP Luke Bradford, Ministry of Health clinical chief advisor rural health Helen MacGregor, and Health New Zealand Te Whatu Ora clinical director primary and community care Sarah Clarke [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/1.%20Barbara%20Fountain%2C%20Luke%20Bradford%2C%20Helen%20MacGregor%20and%20Sarah%20Clarke.jpg?itok=091NETXI)
![Ngāti Porou Oranga specialist GP Elina Pekansaari and Te Nikau Hospital specialist in general practice and rural hospital medicine David Short [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/2.%20Elina%20Pekansaari%20and%20David%20Short.jpg?itok=h5XfSBVM)
![Locum specialist GP Margriet Dijkstra and OmniHealth regional operations manager (southern) Patricia Morais-Ross [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/3.%20Margriet%20Dijkstra%20and%20Patricia%20Morais-Ross.jpg?itok=jkrtRfJC)
![Golden Bay dairy farmer and dairy industry health and safety doctoral student Deborah Rhodes, and Golden Bay Community Health specialist GP Rachael Cowie [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/4.%20Deborah%20Rhodes%20and%20Rachael%20Cowie.jpg?itok=oM0_GcJc)
![Hauora Taiwhenua clinical director rural health Jeremy Webber, Australian College of Rural and Remote Medicine president Rod Martin and Observa Care director of business operations Deborah Martin, the wife of Dr Martin [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/5.%20Jeremy%20Webber%2C%20Rod%20Martin%20and%20Deborah%20Martin%2C%20the%20wife%20of%20Dr%20Martin.jpg?itok=P_aGmX_H)
![Spark Health chief executive John Macaskill-Smith and client director Bryan Bunz [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/6.%20John%20Macaskill-Smith%20and%20Bryan%20Bunz.jpg?itok=5yJvVZ0I)
![Associate dean (rural) Kyle Eggleton, third-year medical student Roselle Winter, and second-year pharmacy student Alina Khanal, all from the University of Auckland [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/7.%20Kyle%20Eggleton%2C%20Roselle%20Winter%20and%20Alina%20Khanal.jpg?itok=RQLd3TEs)
![Health New Zealand Te Whatu Ora clinical editor and specialist in general practice and rural hospital medicine Anu Shinnamon, and Whakarongorau chief clinical officer Ruth Large [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/8.%20Anu%20Shinnamon%20and%20Ruth%20Large.jpg?itok=i5TMswY9)
![Te Kahu Hauora Practice specialist GP Jane Laver and Ngāti Kahungunu ki Tāmaki-nui-a-Rua chief operations manager Tania Chamberlain [Image: NZD]](/sites/default/files/styles/thumbnail_cropped_100/public/2025-05/9.%20Jane%20Laver%20and%20Tania%20Chamberlain.jpg?itok=jtMklaCZ)