For older people and frail people, the long-term benefit of medicines reduces and the potential for harm from adverse effects increases. When the benefit–risk balance changes in this way, medicine review and optimisation are important to simplify the therapeutic regimen, reduce inappropriate medicines and minimise risks. In this article, pharmacist prescriber Linda Bryant uses two case studies to illustrate important considerations during medicine reviews
Aortic stenosis
Aortic stenosis
In this article, Jonathon White writes about the contemporary management of aortic stenosis. He covers the importance of detecting AS early, challenges surrounding diagnosis, indications for treatment, and how management pathways are decided on by the heart team. The definitive treatment for AS – aortic valve replacement – is described in detail
This continuing medical education activity has been endorsed by the RNZCGP and has been approved for up to 1 CME credit for continuing professional development purposes (1 credit per learning hour). To claim your credits, log in to your RNZCGP dashboard to record this activity in the CME component of your CPD programme.
Nurses may also find that reading this article and reflecting on their learning can count as a professional development activity with the Nursing Council of New Zealand (up to 1 PD hours).
At the end of this course, you should be able to:
- Discuss strategies for early diagnosis of aortic stenosis
- Compare treatment pathways for AS
- Describe the surveillance and follow-up of patients with untreated and treated AS
This How to Treat assists with the development of the following domains of competence in the general practice curriculum:
- Domain 1: Communication
- Domain 2: Clinical expertise
- Domain 4: Scholarship
- Domain 5: Context of general practice
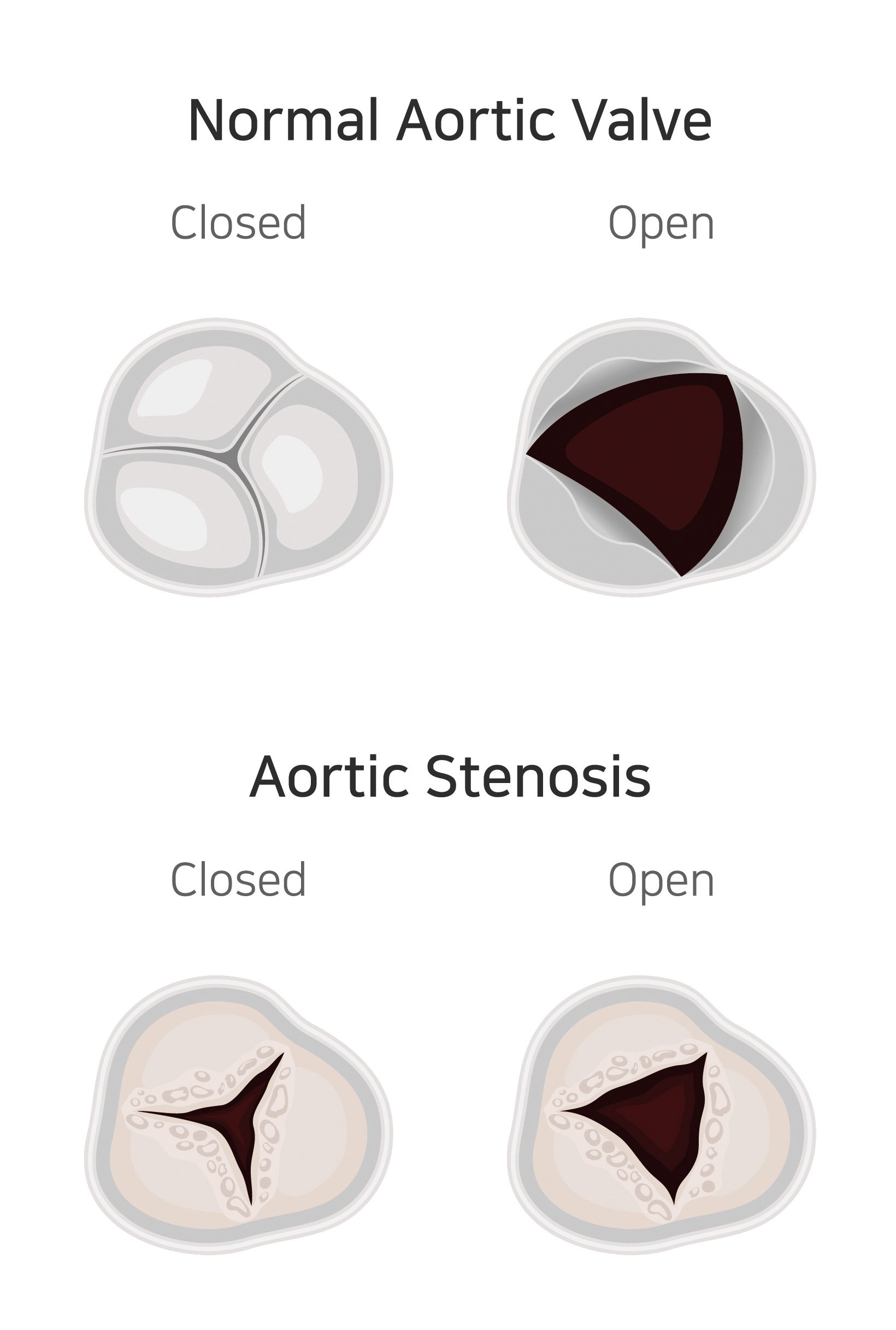
Aortic stenosis is a common degenerative valve lesion with population estimates indicating prevalence increasing to 10 per cent by the age of 80.1 Calcification and restriction of the aortic valve (Figure 1) is a chronic, progressive condition, for which no medical treatment has ever been shown to modify the natural history of the disease.
In addition to age-related AS, other causes include a congenitally malformed aortic valve, rheumatic fever and chest radiation therapy. Risk factors are similar to those for atherosclerosis and include diabetes, obesity, dyslipidaemia, hypertension and chronic kidney disease.
The condition is characterised by a long, clinically latent phase, during which patients are asymptomatic. The risk of complications from AS in this phase is very low, so clinical observation is appropriate. However, once symptoms or left ventricular (LV) dysfunction develop, AS is associated with a dismal prognosis, with 50 per cent of patients dying within one year.2
Given the poor prognosis of AS following the development of symptoms or LV dysfunction, early detection and then close surveillance are imperative.
This article provides top tips to help GPs identify and refer patients who may have AS and then follow up those who undergo treatment.
In the period before symptom onset, the only way of diagnosing AS is auscultation of the heart sounds – the classic description is of a harsh, late-peaking ejection systolic murmur, loudest at the base of the heart. In very severe AS, the second heart sound will be diminished and may not be audible at all.
Given the increasing prevalence of AS in older people, cardiac auscultation should form a part of the assessment for all older patients.
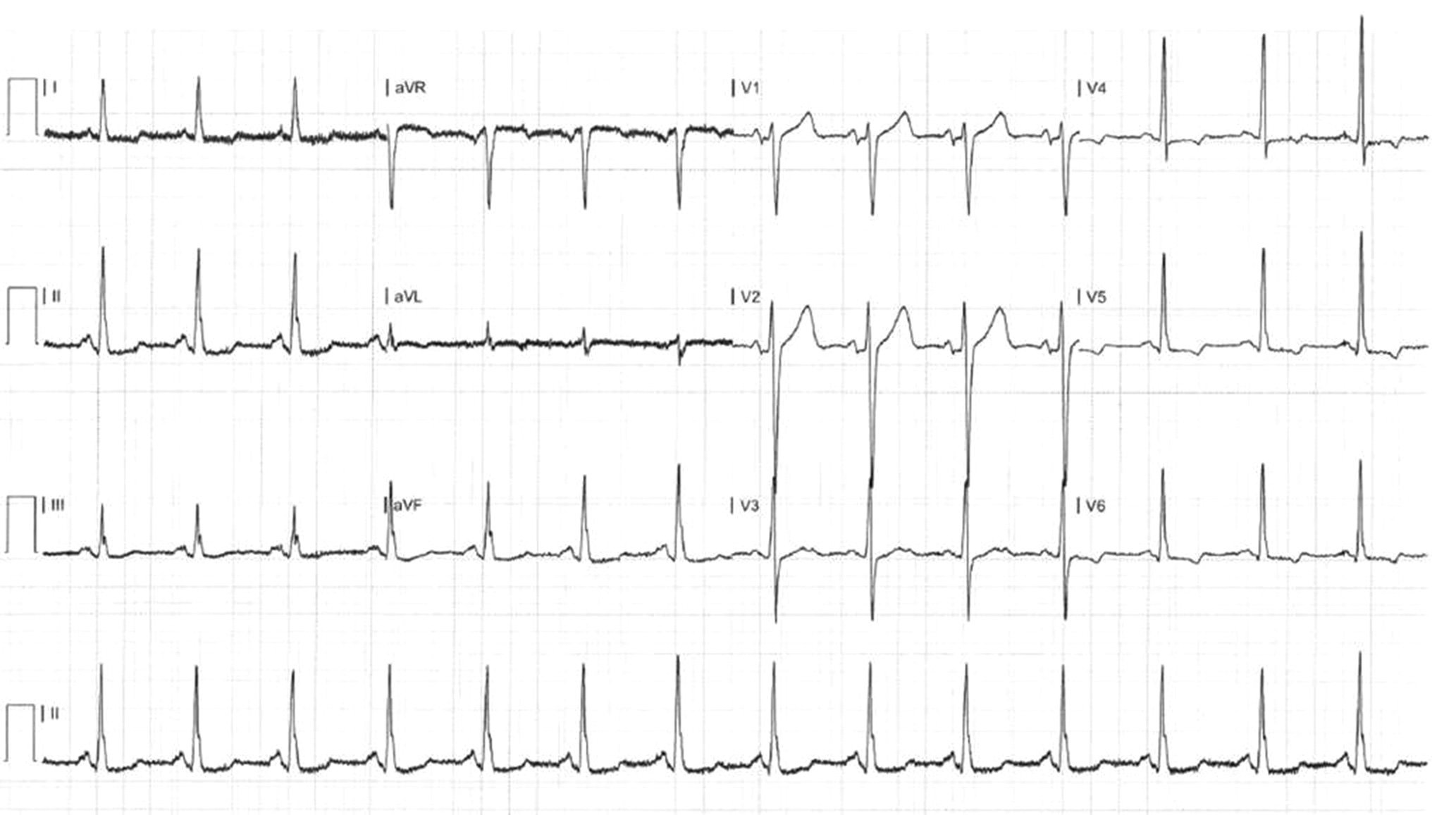
Even in the absence of symptoms, an electrocardiogram may show signs of LV hypertrophy or LV strain (Figure 2).
The classic triad of symptoms is angina, dyspnoea and syncope, but these can manifest late in the course. In older people, symptoms may be non-specific and include fatigue and declining exercise capacity. AS should be actively excluded in patients presenting with heart failure or newly diagnosed LV dysfunction.
Community patients with any of these symptoms, physical signs, or ECG changes should be referred for cardiology assessment and echocardiography.
A summary of the top tips for early detection of AS is shown in the panel.
Echocardiography
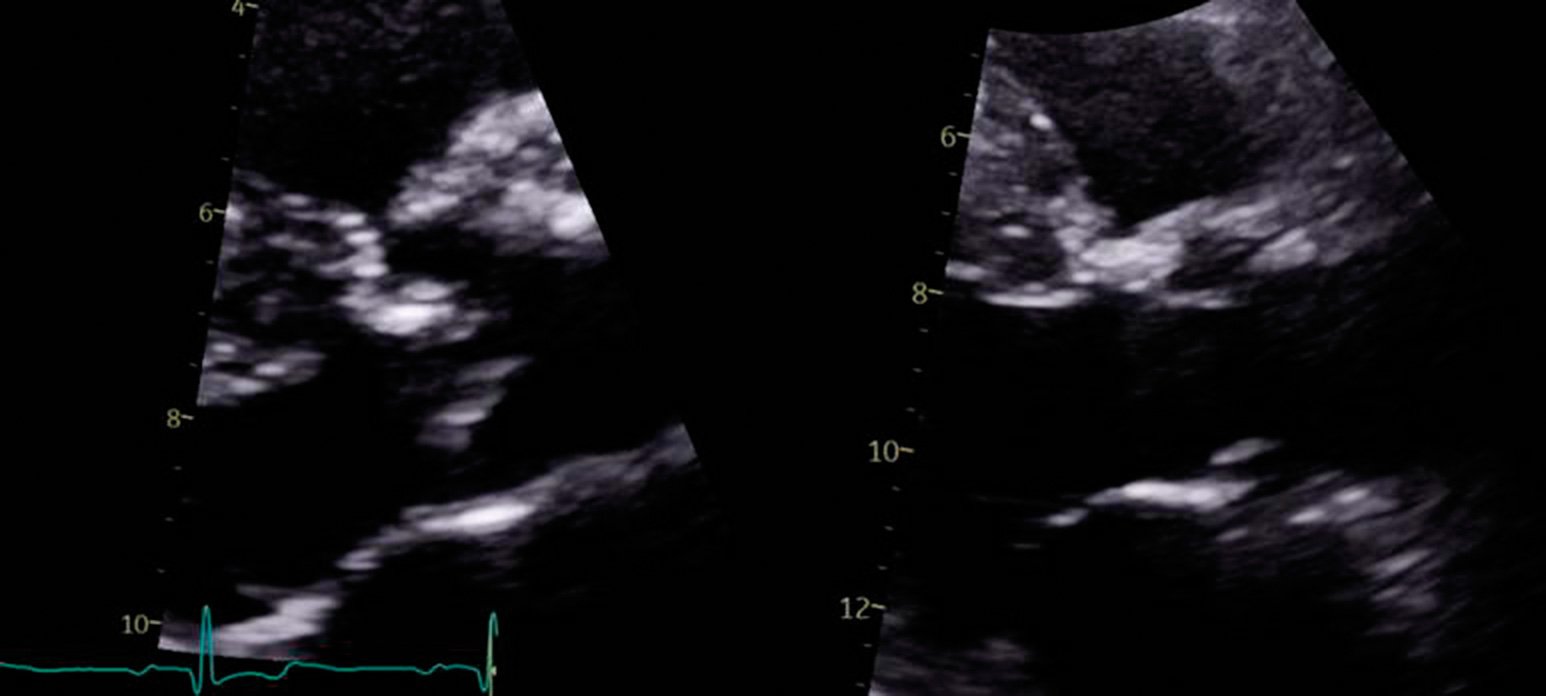
The most important clinical assessment of AS is by echocardiography, and most delays to diagnosis and treatment stem from delays to access to this modality. Transthoracic echocardiography provides two-dimensional assessment of valve morphology – specifically, the degree to which the valve is calcified and restricted (Figure 3).
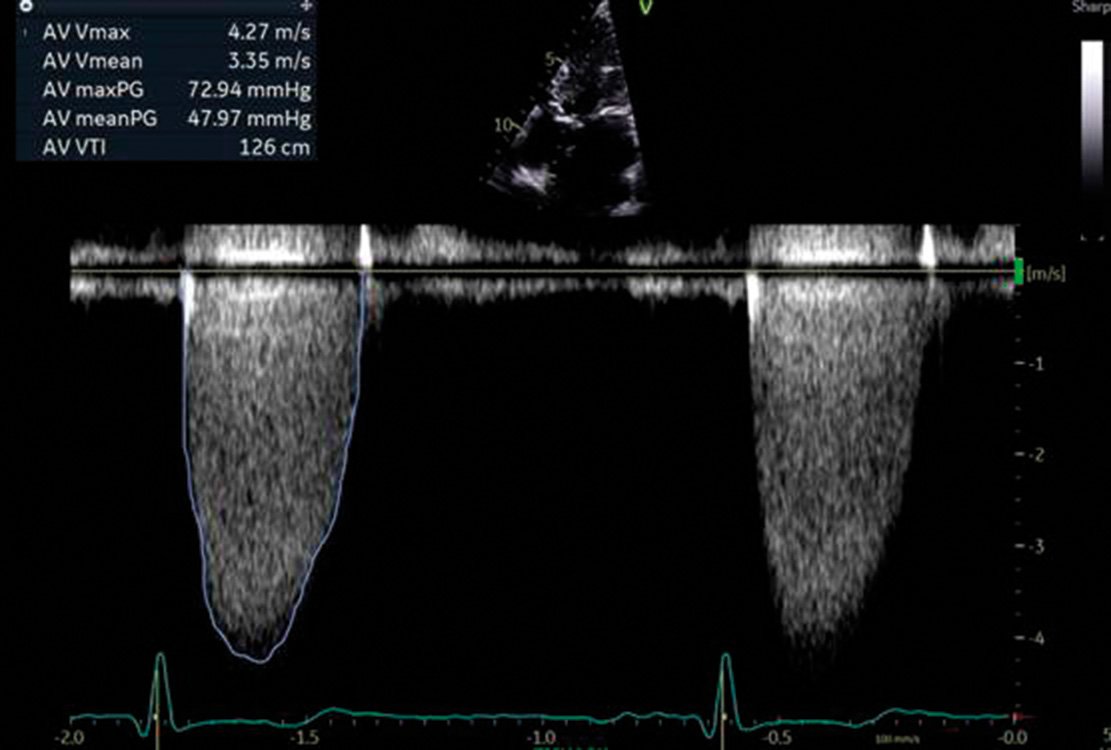
Continuous-wave Doppler assesses the peak velocity of blood through the stenosed valve orifice and, from this Doppler trace, a mean gradient can be derived (Figure 4). An estimated valve area can be derived from integration of Doppler data and two-dimensional measurements.
The grading of AS is a multiparametric one, from which the lesion is graded as mild, moderate or severe.
In the setting of normal flow conditions (normal cardiac output), AS is graded as severe when the peak velocity exceeds 4m/s, the mean gradient exceeds 40mmHg and the valve area falls below 1.0cm2.
These cut-offs may not be met in the setting of low cardiac output, where the heart is unable to generate the forward flow to drive these high velocities. Common conditions that cause this low-flow state are LV dysfunction, other valve lesions (eg, mitral regurgitation) and diastolic dysfunction from hypertension or amyloidosis. With careful assessment, the diagnosis of severe AS can still be made, but this becomes based upon derived valve area and other measures that are independent of flow.
Due to measurement errors and several other limitations of these primary data, all aspects of valve assessment must be carefully considered and matched to the clinical picture of the patient. In this sense, the echocardiographic assessment of AS is very much a balanced, clinical one.
Patients with mild or moderate AS should stay under regular cardiology clinic follow-up. The timing of such follow-up depends on the rate of progression seen in an individual patient. In general, patients with mild AS should be seen every three to five years, with follow-up every one to two years once in the moderate range. Once the patient has severe AS, follow-up should be every six to 12 months, or until the development of symptoms or LV dysfunction.
Indications for treatment
The only definitive treatment for AS is aortic valve replacement (AVR). In order to avoid the morbidity and mortality associated with severe AS, AVR is indicated for:
- symptoms associated with AS
- LV dysfunction.
AVR is also generally considered to be reasonable in asymptomatic patients with AS in the critical range (peak velocity >5m/s and valve area <0.6cm2). Ongoing studies are assessing the role of early treatment of truly asymptomatic severe AS, but this is currently not supported by clinical evidence.
Care must be taken when considering a patient to be truly asymptomatic. Accordingly, guidelines now recommend all patients with severe AS have exercise testing to exclude occult symptoms, exercise-induced hypotension and marked ECG ischaemia.3
In the case of such findings, asymptomatic patients can be reclassified as requiring intervention. In older people where a treadmill test is not practicable, an alternative is a negative B-type natriuretic peptide test before classing them as truly asymptomatic.
Medical therapy of AS
No medical therapy is associated with improved prognosis of AS. However, in those not suitable for AVR or awaiting AVR, medical therapy can improve symptoms or prevent symptom progression while definitive treatment is awaited.
Diuretics to manage heart failure can be cautiously used, but volume depletion must be avoided. Excessive bradycardia is also best avoided, as cardiac output is dependent upon a relatively fixed stroke volume due to the fixed obstruction at the level of the valve.
Patients already established on an ACE inhibitor or angiotensin II receptor blocker can be maintained on one, but these medications are best not initiated in newly diagnosed severe AS due to their potent vasodilatory tendency.
The strength of decision-making for patients with aortic stenosis lies in the recently evolved concept of the heart team
Once AVR is indicated, it should be performed promptly due to the ongoing risk of death, even in the short term. Options now include surgical (open-heart) AVR (SAVR) or transcatheter AVR (TAVR).
Surgical AVR
SAVR can be performed with either a bioprosthetic (tissue) valve or a mechanical one. In the absence of complications, patients typically spend one night in an intensive care unit and five to seven days in hospital following SAVR.
A bioprosthetic valve has the benefit of avoiding anticoagulation (unless otherwise indicated – eg, atrial fibrillation) but has the downside of limited durability. Most surgical bioprosthetic valves can be expected to last between 10 and 20 years, although this varies greatly depending on valve type and patient-related factors.4,5
To avoid the likelihood of valve degeneration requiring further intervention, most patients under age 50 receive a mechanical valve unless there are reasons to avoid anticoagulation with warfarin (eg, family planning, poor compliance, occupational hazards or other patient preferences).
Between ages 50 and 65, treatment choice is often individualised, depending on attitudes to anticoagulation and reintervention. Almost all patients over age 65 will have a bioprosthetic valve, considering that a transcatheter valve can now be placed inside a failed bioprosthesis, avoiding re-operative open-heart surgery.
Transcatheter AVR
TAVR is a percutaneous procedure that has recently emerged as a safe and effective alternative to surgical bioprosthetic valves in carefully selected patients. Because TAVR has a shorter history, there are no data on very-long-term durability of these valves in younger patients.
In order to understand the evolution of this field, it is important to understand that, in clinical practice, patients with AS are classed according to their risk of death within 30 days of open-heart surgery. Patients are stratified into extreme (inoperable), high (>8 per cent risk of death), intermediate (4–8 per cent) and low-risk (<4 per cent) categories.
A typical high-risk patient would be in their 80s with several comorbidities, while intermediate-risk patients are typically those in their 80s with very mild comorbidity or those in their 70s with moderate comorbidity. Low-risk patients are typically those in their 70s or very early 80s with no other medical problems.
A series of randomised trials performed within the last 10 years has looked at the use of TAVR in patients of decreasing surgical risk. These trials began by comparing TAVR with medical therapy alone in patients in whom open-heart surgery was not possible due to extreme surgical risk. These studies showed a large reduction in the risk of death, improvement in symptoms and reduction in heart failure hospitalisation in those treated with TAVR.3
Subsequent trials tested TAVR against SAVR in progressively lower risk cohorts, with TAVR being shown to be safer and more cost-effective than open-heart surgery in high-risk patients.6,7
In intermediate-risk patients, TAVR and SAVR appear to be similar in terms of major adverse events, such as death and stroke, but with a much faster recovery and fewer hospital readmissions with TAVR.8,9
Recent randomised trials in carefully selected low-risk patients showed patients treated with either SAVR or TAVR had excellent outcomes, but with slightly lower incidence of stroke and death with TAVR than with SAVR. In this low-risk group, TAVR had other significant advantages, including fewer cases of atrial fibrillation, shorter hospital stays, fewer subsequent readmissions and improved symptoms and quality of life measures.10,11
One important limitation of these studies on low-risk patients is the currently short follow-up. Given the younger age and lack of comorbidity in these patients, long follow-up takes on much greater importance than in earlier trials.
Presently, transcatheter valves are significantly more expensive than surgical ones, although trial data suggest overall treatment costs still favour TAVR once adjusted for quality of life and other associated costs.12
Making treatment decisions – the heart team
Presently in New Zealand, there is variation in treatment pathways depending on geography and insurance coverage. TAVR is widely held as the treatment of choice for AS in older intermediate, high and extreme-risk patients in whom treatment is not considered futile (futility is covered below). Some DHBs offer TAVR to low-risk patients, while others currently do not.
The strength of decision-making for patients with AS lies in the recently evolved concept of the heart team – a multidisciplinary group of specialists that includes cardiac surgeons, cardiologists, intensive care specialists, nurse specialists and geriatricians.
Other than confirming the diagnosis of severe AS by echocardiography, the most important role of the heart team is to ensure safe and effective treatment of the aortic valve while appropriately managing other heart disease, such as concomitant coronary and mitral valve disease.
The risk of a TAVR procedure is largely dictated by anatomy, as evaluated by pre-procedural CT imaging, which forms the basis of most discussions once TAVR is planned. CT factors that increase procedural risk include calcification of the aortic annulus and heavily calcified iliofemoral arteries.
If arterial access is inadequate for the usual transfemoral approach, alternatives include access to the subclavian/axillary artery or direct surgical access to the aorta or LV apex itself. These are increasingly invasive, so must be considered in the context of comorbidities and overall goals of care, as well as the appropriateness of an open surgical alternative.
The role of the heart team has become increasingly important as younger and more complex patients are considered for treatment. Not only does short-term safety need to be considered but so too the longitudinal care of patients, given the potential for future reintervention. This is quite distinct to the early days of TAVR when the longitudinal care of patients was less important, given the advanced age and comorbid state of many patients in that era.
Although coronary and mitral valve disease may now be treated using percutaneous techniques, in younger, fitter patients, open-heart surgery may serve as a more comprehensive solution, albeit a more invasive one. As these decisions become increasingly complex, the strengths of all members of the heart team are increasingly drawn upon.
Futility – who should not be treated?
Besides the feasibility of TAVR in the treatment of AS, a great deal of what was learned from the early TAVR experience was an understanding of which patients do not thrive despite relief of aortic valve obstruction.
Futility not only refers to major complications from intervention but also the failure to improve in terms of functional status or quality of life.
The most common markers of futility are frailty (poor nutritional or functional status), oxygen dependence (indicating either a very advanced heart failure state or important lung disease), cognitive impairment and major competing non-cardiac comorbidity (eg, malignancy).13 The heart team is instrumental in careful assessment of these factors and establishing appropriate goals of care in such patients.
What to expect from TAVR
Almost all TAVR procedures are now performed via transfemoral access. The technique continues to become less invasive, and many procedures are done under only light sedation with a fully conversant patient.
Using local anaesthetic, a large sheath (up to 6mm in diameter) is inserted into one of the common femoral arteries. This access is closed with suture-based closure devices at the end of the procedure following valve deployment.
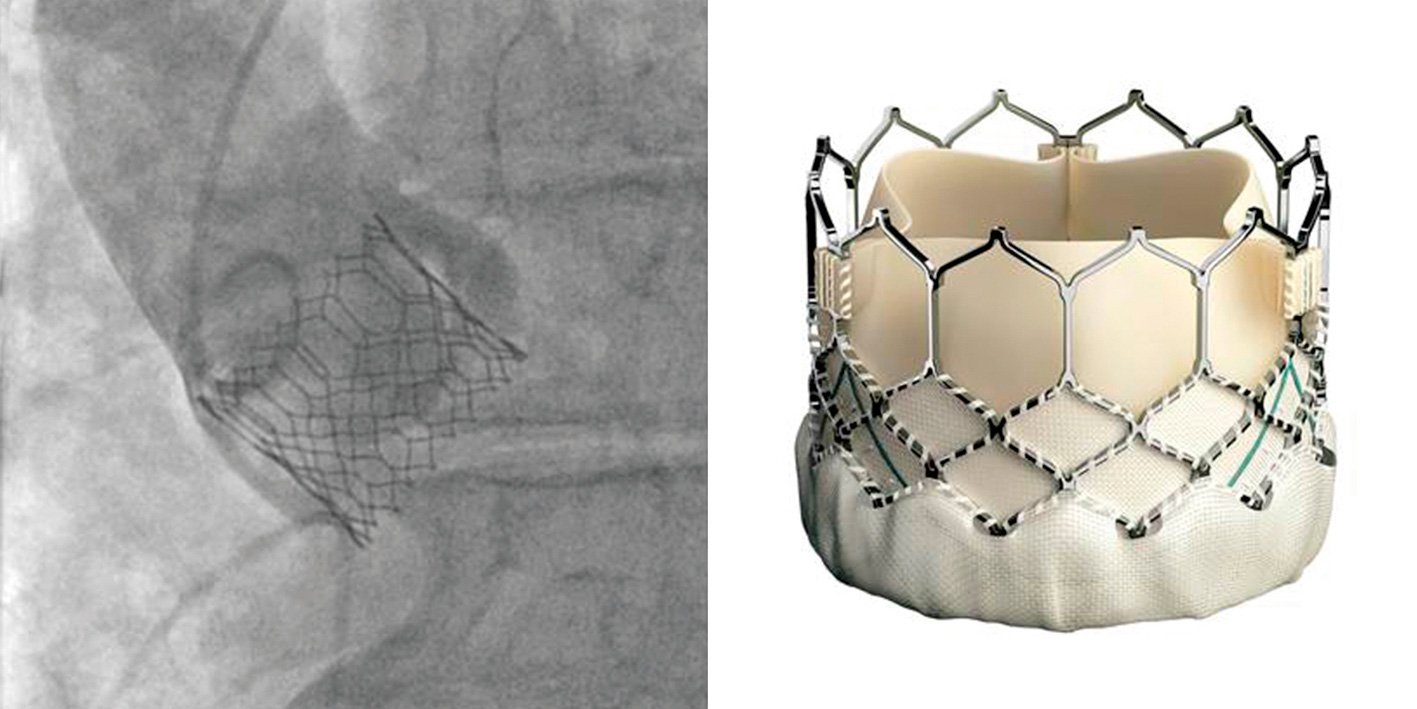
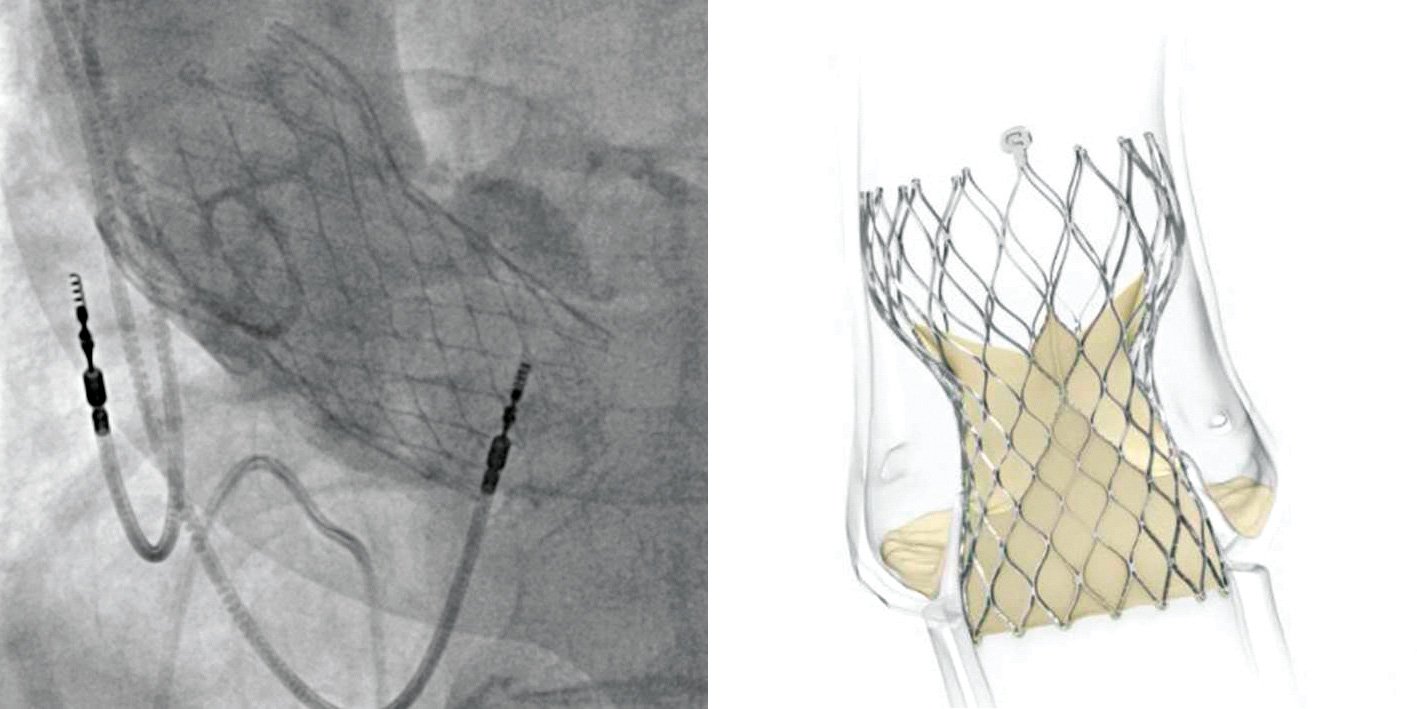
Although multiple TAVR devices are commercially available, the market is dominated by the SAPIEN 3 (Edwards Lifesciences) and Evolut (Medtronic) systems (Figures 5 and 6). These devices have important differences but are both excellent. Their valves are made from bovine and porcine pericardial tissue, respectively.
The SAPIEN 3 is a balloon-expandable device with a slightly larger bore access requirement, while the Evolut system has a higher rate of pacemaker implantation but may be safer in highly calcified anatomy owing to its self-expanding nitinol frame.
Most patients will be discharged on the first post-operative day and can be expected to return to their usual daily activities within just a few days. In those with low-risk CT anatomy, rates of significant complications are now expected to be well below 5 per cent.
Although many patients realise a significant symptomatic improvement immediately, in others, this does take some time, given the chronic effects that AS has on the myocardium, including LV hypertrophy and diastolic dysfunction.
Long-term follow-up after AVR
Follow-up after TAVR is similar to any bioprosthetic AVR. Patients should be treated with aspirin unless at extremely high risk of bleeding. Those who have another indication for either anticoagulation (eg, atrial fibrillation) or clopidogrel (eg, stroke or coronary disease) do not need two antithrombotic agents for the sake of the valve.
Local practice will vary depending on resources available, but it is reasonable to obtain an echocardiogram every one to two years initially, then more frequently as valve degeneration is observed, or if symptoms or other clinical concerns develop.
A mechanical valve requires indefinite anticoagulation with warfarin. Direct or novel oral anticoagulants have been tested as an alternative but have been shown to be associated with higher rates of thromboembolic complications, so they are contraindicated for use in this population.14
Conclusion
Left untreated, severe, symptomatic AS is associated with poor quality of life and prognosis. Early diagnosis by echocardiography and engagement with a heart team are of great importance.
Treatment with AVR is indicated for symptoms of AS or LV dysfunction. Both surgical and transcatheter valve replacements are excellent treatments for AS, and patients can be expected to return to a good quality of life with a low rate of complications after both procedures.
The appropriate treatment choice depends on the patient’s risk of complications from open-heart surgery, CT-defined anatomy, overall functional status and presence of other cardiac disease that requires treatment.
Jonathon White is an interventional cardiologist at The Heart Group, Intra and Auckland Hospital
This 10-question multiple-choice assessment is designed to demonstrate that the provided educational reading has been effective in allowing you to meet the learning objectives of this course. Write down your answers to these questions.
1. What are the characteristic symptoms of the early phase of aortic stenosis?
a. Angina, dyspnoea and syncope
b. Fatigue and declining exercise capacity
c. Tachycardia and oedema
d. None – patients are asymptomatic
2. How are heart sounds typically described in people with AS?
a. A harsh, late-peaking ejection systolic murmur, loudest at the base of the heart
b. A holosystolic murmur, heard best at the apex, with a third heart sound
c. A low-pitched decrescendo-crescendo rumbling diastolic murmur, loudest at the apex
d. A mid-systolic murmur with wide and fixed splitting of the second heart sound
3. Most delays to diagnosis and treatment of AS stem from delays to access to which of the following modalities?
a. Cardiac CT
b. Cardiac MRI
c. Echocardiography
d. Electrocardiography
4. In the setting of normal cardiac output, how is AS graded if the peak velocity of blood through the valve exceeds 4m/s, the mean gradient across the valve exceeds 40mmHg, and the valve area falls below 1.0cm2?
a. Mild
b. Moderate
c. Severe
d. Critical
5. The only definitive treatment for AS is aortic valve replacement. True or false?
a. True
b. False
6. What is the appropriate clinical response for a patient with truly asymptomatic AS?
a. Aortic valve replacement
b. Close clinical observation by the cardiology clinic
c. Treatment with a beta-blocker
d. Treatment with an ACE inhibitor or angiotensin II receptor blocker
7. Treatment with aortic valve replacement is indicated under which TWO of the following scenarios?
a. AS with oxygen dependence due to advanced heart failure
b. AS with symptoms of AS or left ventricular dysfunction
c. Asymptomatic severe AS
d. Asymptomatic severe AS for at least 12 months
e. Asymptomatic severe AS with abnormalities found upon exercise stress testing
8. What is an advantage of mechanical valves over bioprosthetic valves for aortic valve replacement?
a. Avoiding anticoagulation
b. Avoiding open-heart surgery
c. Long-term durability
d. Lower rate of complications
9. Which TWO statements regarding transcatheter aortic valve replacement (TAVR) are correct?
a. A transcatheter valve can be placed inside a failed bioprosthetic valve
b. Surgical AVR has better outcomes than TAVR in younger patients without comorbidities
c. TAVR has a long history of use in younger patients without comorbidities
d. The risk of a TAVR procedure is largely dictated by CT-defined anatomy
10. Following TAVR, how often should patients be followed up with echocardiography?
a. Every six months
b. Every year, but only once symptoms or other clinical concerns develop
c. Every one to two years, then more frequently as valve degeneration is observed
d. Every three to five years, then more frequently as valve degeneration is observed
Write down your answers to these questions. Then, to check your answers and record your score, click here.
You can use the Capture button below to record your time spent reading and your answers to the following learning reflection questions:
- Why did you choose this activity (how does it relate to your professional development plan learning goals)?
- What did you learn?
- How will you implement the new learning into your daily practice?
- Does this learning lead to any further activities that you could undertake (audit activities, peer discussions, etc)?
We're publishing this Educate article as a FREE READ so it can be read and shared more widely. If you would like to access comprehensive primary care education, and support us – subscribe here
1. Eveborn GW, Schirmer H, Heggelund G, et al. The evolving epidemiology of valvular aortic stenosis. The Tromsø study. Heart 2013;99(6):396–400.
2. Leon MB, Smith CR, Mack M, et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med 2010;363(17):1597–607.
3. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021;143(5):e72–227.
4. Foroutan F, Guyatt GH, O'Brien K, et al. Prognosis after surgical replacement with a bioprosthetic aortic valve in patients with severe symptomatic aortic stenosis: systematic review of observational studies. BMJ 2016;354:i5065.
5. Bourguignon T, Bouquiaux-Stablo AL, Loardi C, et al. Very late outcomes for mitral valve replacement with the Carpentier-Edwards pericardial bioprosthesis: 25-year follow-up of 450 implantations. J Thorac Cardiovasc Surg 2014;148(5):2004–11.e1.
6. Adams DH, Popma JJ, Reardon MJ, et al. Transcatheter aortic-valve replacement with a self-expanding prosthesis. N Engl J Med 2014;370(19):1790–98.
7. Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011;364(23):2187–98.
8. Reardon MJ, Van Mieghem NM, Popma JJ, et al. Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. N Engl J Med 2017;376(14):1321–31.
9. Leon MB, Smith CR, Mack MJ, et al. Transcatheter or surgical aortic-valve replacement in intermediate-risk patients. N Engl J Med 2016;374(17):1609–20.
10. Mack MJ, Leon MB, Thourani VH, et al. Transcatheter aortic-valve replacement with a balloon-expandable valve in low-risk patients. N Engl J Med 2019;380(18):1695–705.
11. Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter aortic-valve replacement with a self-expanding valve in low-risk patients. N Engl J Med 2019;380(18):1706–15.
12. Baron SJ, Wang K, House JA, et al. Cost-effectiveness of transcatheter versus surgical aortic valve replacement in patients with severe aortic stenosis at intermediate risk. Circulation 2019;139(7):877–88.
13. Lindman BR, Alexander KP, O'Gara PT, et al. Futility, benefit, and transcatheter aortic valve replacement. JACC Cardiovasc Interv 2014;7(7):707–16.
14. Eikelboom JW, Connolly SJ, Brueckmann M, et al. Dabigatran versus warfarin in patients with mechanical heart valves. N Engl J Med 2013;369(13):1206–14.