For older people and frail people, the long-term benefit of medicines reduces and the potential for harm from adverse effects increases. When the benefit–risk balance changes in this way, medicine review and optimisation are important to simplify the therapeutic regimen, reduce inappropriate medicines and minimise risks. In this article, pharmacist prescriber Linda Bryant uses two case studies to illustrate important considerations during medicine reviews
The role of susceptibility reporting in antimicrobial stewardship, but what is ‘I’?
+Practice
In print
FROM THE LAB
The role of susceptibility reporting in antimicrobial stewardship, but what is ‘I’?
Wednesday 21 July 2021, 12:18 AM
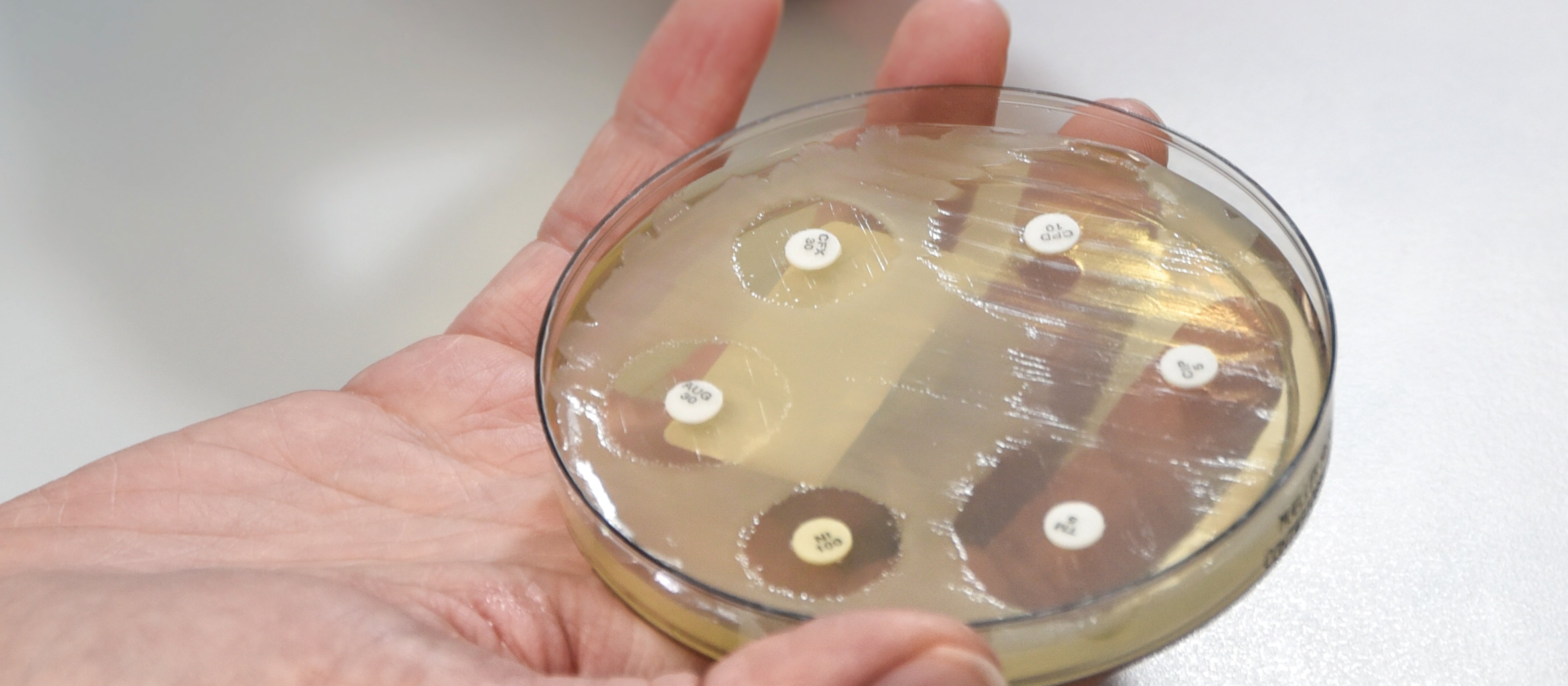
A zone of no growth around an antibiotic-impregnated disc demonstrates a susceptible organism \
Manual techniques for antimicrobial susceptibility testing are relatively simple, but interpretation and reporting of results are less so. This article explains the meaning of “I”, the importance of dosing, and the role of microbiology in antimicrobial stewardship
Juliet Elvy
This article has been endorsed by the RNZCGP and has been approved for up to 0.25 CME credits for the General Practice Educational Programme and conti
Kia ora and welcome to New Zealand Doctor Rata Aotearoa
Not a subscriber? Unlock this article by subscribing here.
References
- European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 11.0, 2021. www.eucast.org/clinical_breakpoints
- Thomas MG, Smith AJ, Tilyard M. Rising antimicrobial resistance: a strong reason to reduce excessive antimicrobial consumption in New Zealand. N Z Med J 2014;127(1394):72–84.
- Williamson DA, Roos RF, Verrall A. Antibiotic consumption in New Zealand, 2006–2014. Porirua, NZ: ESR; 2016. https://bit.ly/3w6XQBJ
- Thomas M, Tomlin A, Duffy E, et al. Reduced community antibiotic dispensing in New Zealand during 2015-2018: marked variation in relation to primary health organisation. N Z Med J 2020;133(1518):33–42.
- Barlam TF, Cosgrove SE, Abbo LM, et al. Implementing an antibiotic stewardship program: Guidelines by the Infectious Diseases Society for America and the Society for Healthcare Epidemiology of America. Clin Infect Dis 2016;62(10):e51–77.
- Gardiner SJ, Pryer JA, Duffy EJ. Survey of antimicrobial stewardship practices in public hospitals in New Zealand district health boards. N Z Med J 2017;130(1458):27–41.
- Green JK, Gardiner SJ, Clarke SL, et al. Antimicrobial stewardship practice in New Zealand’s rural hospitals. N Z Med J 2018;131(1481):16–26.
- Langford BJ, Seah J, Chan A, et al. Antimicrobial stewardship in the microbiology
laboratory: Impact of selective susceptibility reporting on ciprofloxacin utilization
and susceptibility of gram-negative isolates to ciprofloxacin in a hospital setting. J
Clin Microbiol 2016;54:2343–47.
- Coupat C, Pradier C, Degand N, et al. Selective reporting of antibiotic susceptibility data improves the appropriateness of intended antibiotic prescriptions in urinary tract infections: a case vignette randomised study. Eur J Clin Microbiol Infect Dis 2013;32(5):627–36.
- Tan TY, McNulty C, Charlett A, et al. Laboratory antibiotic susceptibility reporting and antibiotic prescribing in general practice. J Antimicrob Chemother 2003;51(2):379–84.
- McNulty C, Lasseter GM, Charlett A, et al. Does antibiotic susceptibility reporting influence primary care prescribing in urinary tract infection and other infections? J Antimicrob Chemother 2011;66(6):1396–404.
- Costelloe C, Metcalfe C, Lovering A, et al. Effect of antibiotic prescribing
in primary care on antimicrobial resistance in individual patients: systematic review
and meta-analysis. BMJ 2010;340:c2096.
- European Centre for Disease Prevention and Control. Proposals for EU guidelines on the prudent use of antimicrobials in humans. Stockholm, Sweden: ECDC; 2017. https://bit.ly/368JUMQ
- Royal College of Pathologists of Australasia. Selective Reporting of Antimicrobials in Australia. Surry Hills, NSW: RCPA; 2019. https://bit.ly/3jwcMXm
- Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis 2014;58:980–83.
- Barenfanger J, Arakere P, Dela Cruz R, et al. Improved outcomes associated with limiting identification of Candida spp. in respiratory secretions. J Clin Microbiol 2003;41(12):5645–49.
- Choosing Wisely. Choosing Wisely Recommendations on the Wise Use of Antibiotics November 2020. https://bit.ly/361HIXC



